30
Apr
In modern biomedical engineering, titanium and its alloys have evolved from being an optional material to becoming the structural backbone of medical implants. From orthopedic devices and dental implants to high-end surgical components, titanium alloys define the performance limits of implantable systems through a unique combination of material properties and advanced manufacturing compatibility.

The dominance of titanium alloys originates from their ability to remain stable and cooperative within the human body.
The first key factor is biocompatibility and surface passivation. When exposed to body fluids, titanium rapidly forms a dense and stable oxide layer:
Ti + O₂ → TiO₂
This naturally formed TiO₂ film provides excellent corrosion resistance and effectively prevents ion release into surrounding tissues, minimizing the risk of inflammation and rejection. Additionally, its surface characteristics promote cell adhesion, laying the foundation for successful osseointegration.
The second advantage lies in mechanical compatibility. Titanium alloys have an elastic modulus of approximately 110 GPa, significantly lower than stainless steel or cobalt-chromium alloys, and closer to that of human bone (10–40 GPa). This reduces the risk of stress shielding, allowing bone tissue to maintain physiological loading and preventing long-term bone resorption.
Different titanium grades are selected based on specific clinical and mechanical requirements.
Commercially pure titanium (TA1–TA4): offers excellent formability and corrosion resistance, making it suitable for low-load applications such as dental implants and pacemaker housings.
Ti-6Al-4V ELI (TC4 ELI): is the industry standard for load-bearing applications. With strict control of interstitial elements such as oxygen, iron, and hydrogen, this alloy achieves superior fracture toughness and fatigue resistance. It is widely used in orthopedic implants including bone plates, screws, and artificial joints.
From a regulatory perspective, medical titanium must comply with standards such as GB/T 13810, ensuring strict control over microstructure, inclusions, and internal defects—critical factors for long-term implant safety.
While titanium alloys offer outstanding in-service performance, they are notoriously difficult to machine.
Key challenges include:
In medical manufacturing, these challenges are even more critical due to stringent requirements such as micron-level tolerances, ultra-smooth surfaces, and zero thermal damage.
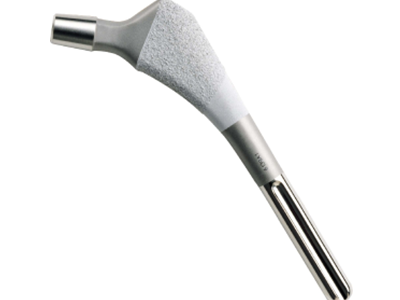
Superhard cutting tools play a crucial role in improving machining efficiency and stability when processing titanium alloys.
To optimize performance, machining strategies typically involve high-pressure coolant systems, low depth of cut with higher feed rates, and proper edge preparation.

Grinding is often the final and most critical step in medical implant manufacturing, directly affecting functional performance and longevity.
To prevent thermal damage and ensure surface integrity, it is essential to use sufficient coolant, optimize grinding parameters, and maintain proper dressing conditions.

With the rise of additive manufacturing, titanium alloys are increasingly used to produce porous implants that better mimic natural bone.
These structures reduce stiffness mismatch, promote bone ingrowth, and improve long-term implant stability. However, they also introduce new machining challenges, requiring advanced finishing processes such as micro-grinding and polishing.
Medical titanium alloys are more than just materials—they are the foundation that connects material science, precision manufacturing, and clinical success. From surface passivation to advanced machining with superhard tools, every step plays a vital role in determining implant performance.
As additive manufacturing and personalized medicine continue to evolve, titanium alloys will remain at the forefront, supported by continuous innovation in cutting and grinding technologies.









